The transition to renewable energies is dependent on increasing the capacity for energy storage. Chemical storage in the form of ammonia is a promising solution, and the stored energy is recovered through combustion. This research examines the effect of plasma chemistry on ammonia combustion,
- Ignition control: expansion of flammability limits and shortening of ignition delay time,
- Reforming: hydrogen and other reforming products for dual-fuel applications,
- Emission control: prevention of NOx formation and unburnt fuel emissions from incomplete combustion.
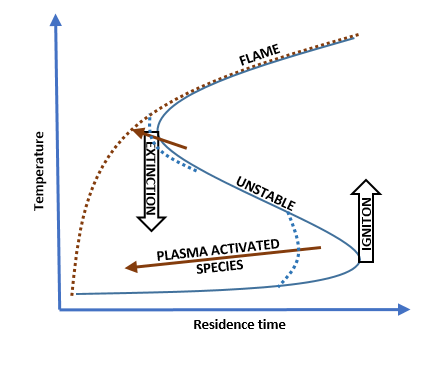
Figure 1
The effect of a high frequency nanosecond pulsed discharge on ignition
A non-thermal plasma discharge at a high frequency creates radicals at low temperatures, initiating alternative reaction pathways. Figure 2 illustrates how a pulsed discharge in the early stages of ignition quickly creates radicals and advances ignition, when compared to a non-pulsed case. A discharge can also promote flame stability and ignition at low temperatures in a similar manner.
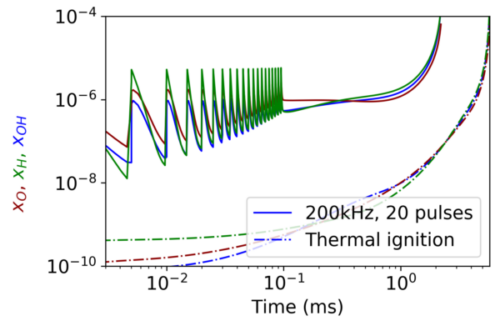
Figure 2
